pH & Skin Cleansers

There are many skin conditions we see every day in our clinics where the practices of our clients or patients have unwittingly been contributing to the development or acceleration of their conditions by using inappropriate cleansers and practices. I am talking about the destruction of the acid mantle.
When we are conducting our initial consultations, the importance of the client bringing their cleansers with them cannot be understated. A major contributing cause of their condition could well be in their daily use products.

As skin treatment professionals, we understand that the skin is the first line of defence against all elements, such as microorganisms, atmospheric conditions, and pollutants.
What we sometimes forget, and the client/patient probably does not fully appreciate, is the importance of the acid mantle, the thin, slightly acidic hydrolipidic film on the surface of the skin that provides the very first line of protection.
The acid mantle plays a critical role as an integral part of the barrier function of the stratum corneum, and consequently, any impairment of its correct functioning will have a knock-on effect to other cells and systems below the surface, and more specifically, the correct functioning of the skins immune system.
It has been observed that when skin surface pH has been allowed to increase too much, it creates susceptibility to bacterial skin infections, skin damage and disease.
Before discussing what kind of things can adversely affect the pH of acid mantle, let us briefly review its properties and characteristics.
The acid mantle is a complex fluid entire formed by substances excreted from the sudiferous and sebaceous glands, epidermic lipids and NMF and is an end result of the skins own metabolism.
The acid mantle contains lactic acid and various amino acids from sweat, free fatty acids from sebum, and amino acids and pyrrolidine carboxylic acid from the cornification process of skin.

The increased enzyme activity of phospholipase A2 (The upstream regulators of many inflammatory processes) has been shown as one of the acid mantles trigger formation processes.
The acid mantle normally has a pH of between 4 and 6.5 in healthy people, with an ideal of 5.5. (pH often varies in different areas of the skin) It is a slightly acidic environment in which protective species of micro flora/bacteria reside. In addition to the inhibition of harmful bacteria, the water/oil emulsion properties make the skin less permeable to water and other polar compounds and regulate trans-epidermal flow/loss. (TEWL)
The acid mantles primary function is to protect the skin from bacterial and fungal infections, provide indirect protection against invasion by microorganisms, and direct protection against alkaline substances. (Via its alkali neutralizing capacity)
The acid mantle also supports the formation and maturation of epidermal lipids and as a result, the maintenance of the barrier function.
The disrupted acid mantle
If the acid mantle becomes disrupted, damaged, or loses its acidity, the skin becomes more susceptible to damage and infection. Washing skin with high alkalinity soaps or detergents can cause the temporary loss of acid mantle, and repetitive washing will change the skin pH, affecting the stratum corneum and barrier functions.
Facial areas are generally not washed as frequent as hands and other areas, but in most cases at least twice a day. Dependent on how alkaline the cleanser used is and how healthy (normal sudiferous and sebaceous flows) the skin is, the return to normal pH can vary between three and fourteen hours. (By which time it will be most likely under further assault from another washing)
Continued washing with alkaline substances can effectively strip the acid mantle for up to 16 hours or 2/3 of each day, and over extended periods, the skin pH is not able to return to normal for sufficient time, and this is where it can affect localised skin health. Any increase in skin surface pH will encourage abnormal bacterial growth.

Soaps & detergents
It is widely understood that soaps generally make the skin more alkaline than synthetic detergents. The irritancy potential of cleansing agents is dependent on a number of factors, which include pH, and soaps are known to increase skin surface pH. Acidic cleansers are less irritating than neutral or alkaline ones, and people susceptible to dry skin are usually advised to use acidic cleansers.
Cleaning agents with slightly acidic or neutral pH, non-ionic surfactants, may be preferable for clients/patients who are at increased risk for irritating skin reactions. Consequently, it is a professional duty to advise patients with skin conditions to choose a mild cleaning agent with a low pH. Even minor differences in the pH of skin cleansing preparations can be important to the integrity of the skin surface. This should be taken into account when determining the optimal soap.
Below are the test results of a selection of products brought to one of my cosmetic chemistry seminars. There are some household cleansers in the group for comparative purposes.
| pH | Brand/Product | Type |
| 3.2 | J&J Clean & Clear Blackhead clearing cleanser | Cleanser |
| 4.6 | Rodan & Fields Proactiv Solution | Renewing cleanser |
| 4.8 | Nimue Youth Facial wash | Wash |
| 5.6 | ASAP daily facial cleanser | Cleanser |
| 5.6 | Ajax spray & wipe | Household cleanser |
| 5.8 | Olay daily facials | Cleanser |
| 6.4 | Soft as soap | Liquid soap |
| 6.6 | Environ Interactive | Gel Cleanser |
| 6.7 | Cetaphil | Cleanser |
| 6.7 | Natures Organics | Foaming face wash |
| 6.8 | Blast Ultraplus | Dishwash concentrate |
| 6.8 | Mila dpiz | Cleansing gel |
| 6.8 | Palmolive Softwash | Liquid soap |
| 6.9 | Dove beauty bar | Bar soap |
| 7.2 | Bubble magic | Bubble bath |
| 8.0 | Environ B active Sebuwash | Facewash |
| 8.0 | Redwin Sorbolene | Bar soap |
| 8.0 | Gatineau nutriactive | Cleanser |
| 8.2 | Clean & clear | Facial wash |
| 9.5 | Palmolive Aroma crme | Bar soap |
| 9.6 | Palmolive Naturals | Bar soap |
| 9.6 | Radox herbal bath | Bath salts |
| 9.7 | J&J Baby soap | Bar soap |
| 9.9 | Honey kids soap | Childrens soap |
| 10.0 | Nelum Sandalwood | Bar soap |
| 10.0 | Lux supreme cream | Bar soap |
| 10.3 | Simple soap | Bar soap |
| 10.3 | Sunlight pure soap | Bar soap |
| 10.3 | AMBR (Hotel brand) | Oatmeal Bar soap |
| 10.5 | Nivea crme bar | Bar soap |
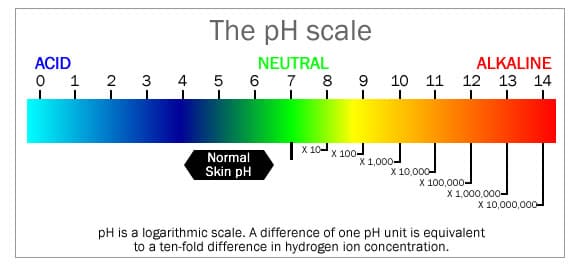
A word on pH
Acidic and alkaline are two extremes that describe a chemical property of a substance. A substance that is neither acidic nor alkaline is neutral. (Such as Pure water at a neutral pH 7.0)
The pH scale measures how acidic or alkaline a substance is, with the pH scale ranging from 0 to 14.
A pH of 7 is neutral. A pH less than 7 is considered acidic, and a pH greater than 7 is considered alkaline. The pH scale is logarithmic and as a result, each whole pH value below 7 is ten times more acidic than the next higher value. For example, pH 4 is ten times more acidic than pH 5 and 100 times (10 times 10) more acidic than pH 6. The same applies for pH values above 7, each of which is ten times more alkaline than the next lower whole value. For example, pH 10 is ten times more alkaline than pH 9 and 100 times (10 times 10) more alkaline than pH 8. The chart above illustrates this.
Our responsibility
Given that the effectiveness of low pH soaps and cleansers has been well documented, one could be forgiven for thinking that home care product manufacturers would meet the market. However, there is a surprisingly limited number of low- pH soaps and cleansers readily available on our supermarket shelves.
Most popular “soaps”, with a few exceptions, are in the pH of 9.5 to 10.5 range. This pH is inherent to the formula, which is usually a sodium soap of fatty acids. Many formulas that have a neutral pH are described as a ” syndet,” and chemically, these are not soaps, but a synthetic detergent in a bar form (as the acronym suggests).
When we have clients who are quite obviously scrubbing away their acid mantle, or have a delicate or damaged mantle, it is our duty to advise alternatives.
Unless we, as skin care professionals, educate our clients about the importance of low- pH cleansers and what is available, they prpbably won’t know to look beyond the most common advertised soaps on the market.
Other Factors that Affect skin pH
It must be mentioned that there are a number of diseases (skin and systemic) that are known to cause an increase in skin surface pH, and consequent skin conditions similar to overuse of alkaline products. During your consultation, you would have been made aware of any potential medical causes for the conditions observed. Skin conditions that can cause the increased pHs phenomenon include:
- Eczema
- Contact dermatitis
- Atopic dermatitis
Acute eczema with erosion can cause skin surface pH to shift from normal to between 7.3 – 7.4.
The entire skin surface pH is increased on skin of people with atopic dermatitis. This increased skin pH contributes to Staphylococcus aureus colonization, which can play a role in the beginning of atopic dermatitis, discoid eczema, and infective dermatitis as a superantigen.
Systemic diseases that can cause an increase in skin surface pH include:
- Diabetes
- Chronic renal failure
- Cerebrovascular disease
The explanation for the elevated pH in these diseases is not fully understood, however is possibly related to low levels of phospholipase (enzyme that hydrolyzes phospholipids into fatty acids and other lipophilic substances) and to autonomic dysfunction causing abnormal sweat secretion.
Have you seen Florence’s book about Cosmetic Chemistry? Take a look here.
Stay Notified
Sign up for news on topics of interest that will help you grow your career and our new releases.
